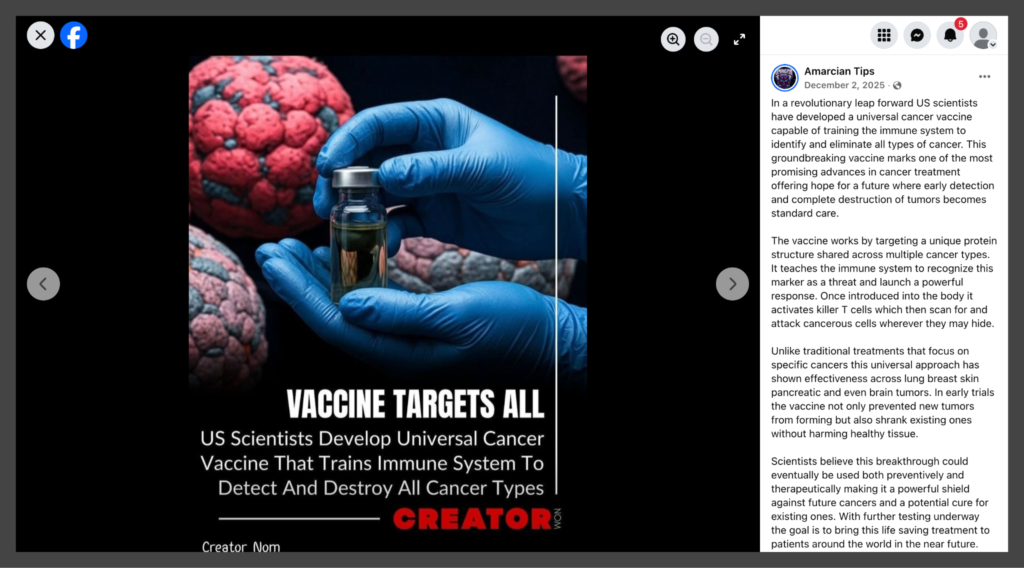
A claim circulating widely on social media platforms states that U.S. scientists have developed a “universal cancer vaccine” capable of training the immune system to recognize and destroy all forms of cancer. The claim is often framed as a major medical breakthrough that can both eliminate tumors and prevent new ones from forming. However, a review of available scientific evidence and official research publications shows that this claim is misleading.
Social Media Posts
Viral posts typically assert that scientists have developed a vaccine that works across all types of cancer. Some versions claim the vaccine can shrink tumors without harming healthy tissue.

Fact Check
What the research actually shows
The claim is based on real research, particularly from a team at the University of Florida. According to an official university release, scientists developed an experimental mRNA-based vaccine designed to stimulate the immune system broadly rather than target a specific cancer marker. The study demonstrated that when this vaccine was combined with immunotherapy drugs, it significantly enhanced anti-tumor responses in mice.
Crucially, this approach works by “waking up” the immune system, mimicking how the body reacts to viral infections, rather than teaching immune cells to recognize a universal cancer-specific marker. Researchers explicitly described their findings as a step toward a potential universal cancer vaccine, not a finished product. As noted in the University of Florida report, the results bring scientists “one step closer” to that goal, indicating early-stage progress rather than completion.
Similarly, reporting from ScienceDaily confirms that the vaccine showed strong anti-tumor effects in mouse models when paired with immune checkpoint inhibitors, but emphasizes that the findings are preclinical.
Preclinical evidence vs. human application
A key limitation is that much of the evidence comes from laboratory and animal studies. Preclinical results, especially in mice, do not automatically translate into safe or effective treatments in humans. Experts consistently stress that further clinical trials are required to evaluate safety, dosing, and real-world effectiveness.
Even related human data remains preliminary. For example, observational research involving cancer patients receiving mRNA COVID-19 vaccines alongside immunotherapy found improved survival outcomes, but these findings require validation in randomized trials before firm conclusions can be drawn.
Researchers themselves caution that these results are early-stage. As one pediatric oncologist at University of Florida noted, the concept of a universal cancer vaccine remains hypothetical and must be confirmed through future clinical studies before it can be considered a viable treatment. (Source)
The viral claim does not fully reflect either the mechanism described by researchers or how early the work still is. At this time, there is no single vaccine shown in humans to work across all cancer types. Cancer includes many distinct conditions with different genetic and biological features, which is one reason many current cancer vaccines are tailored to specific tumors rather than broadly applicable.
Evidence from early clinical trials (mRNA-4359 example)
It is important to distinguish this from the University of Florida research described earlier. The Florida study refers to a separate experimental vaccine tested only in mice, whereas mRNA-4359 is a different mRNA-based cancer vaccine currently being tested in humans in early-stage clinical trials.
Specifically, mRNA-4359 is being evaluated in an ongoing Phase 1/2 study involving patients with advanced solid tumors, where the primary objective is to assess safety, tolerability, and appropriate dosing rather than treatment effectiveness . Early interim data presented in 2024-2025 showed that the vaccine was generally well tolerated and capable of triggering immune responses, including activation of T cells targeting cancer-related proteins such as PD-L1 and IDO1 .
However, these findings remain preliminary. The trials involve small patient groups and are designed mainly to determine safety, not to demonstrate that the vaccine can cure cancer or work across all tumor types. Researchers explicitly caution that results should be interpreted carefully due to the limited sample size and early stage of development .
In short, while mRNA-4359 represents early human testing, it is not the same as the mouse-based “universal vaccine” study, and neither provides evidence of a proven, broadly effective cancer vaccine.
(Source: MedPath, ClinicalTrials.gov, ecancer)
Conclusion
The claim that U.S. scientists have developed a universal cancer vaccine is misleading. While researchers have made promising progress with experimental mRNA vaccines, particularly in animal studies and early clinical trials, there is currently no proven universal cancer vaccine for humans. The existing evidence reflects early-stage research that may inform future treatments, but it does not support claims of a ready, all-purpose cure for cancer.